Built to Perform. Built to Endure.
It is a core part of the platform.
Every design decision was made with one objective in mind: to support sterile injectable manufacturing that remains controlled, defensible, and scalable as programs mature and expectations increase.
Designed From First Principles
Our facility was designed intentionally, not inherited or retrofitted.
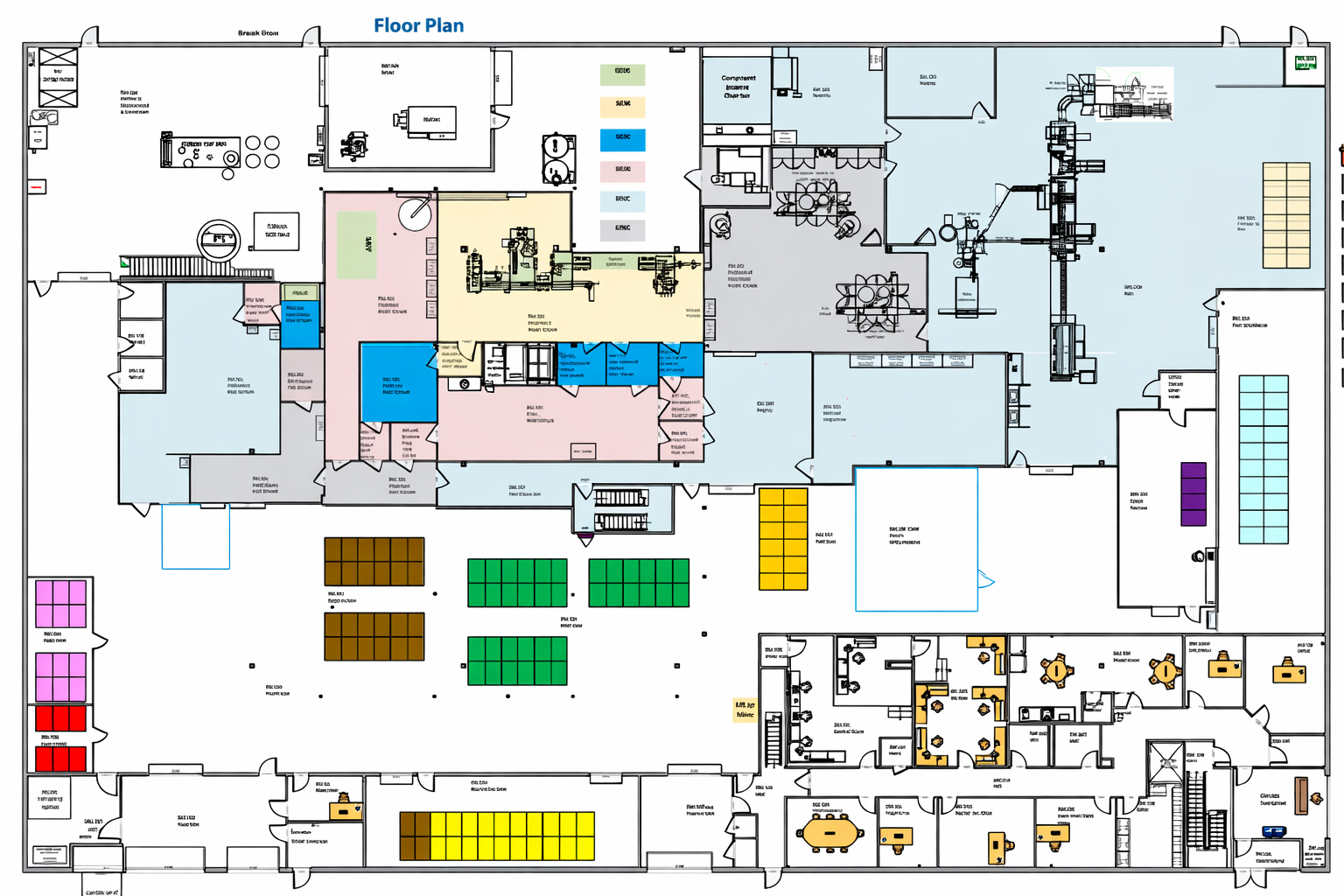
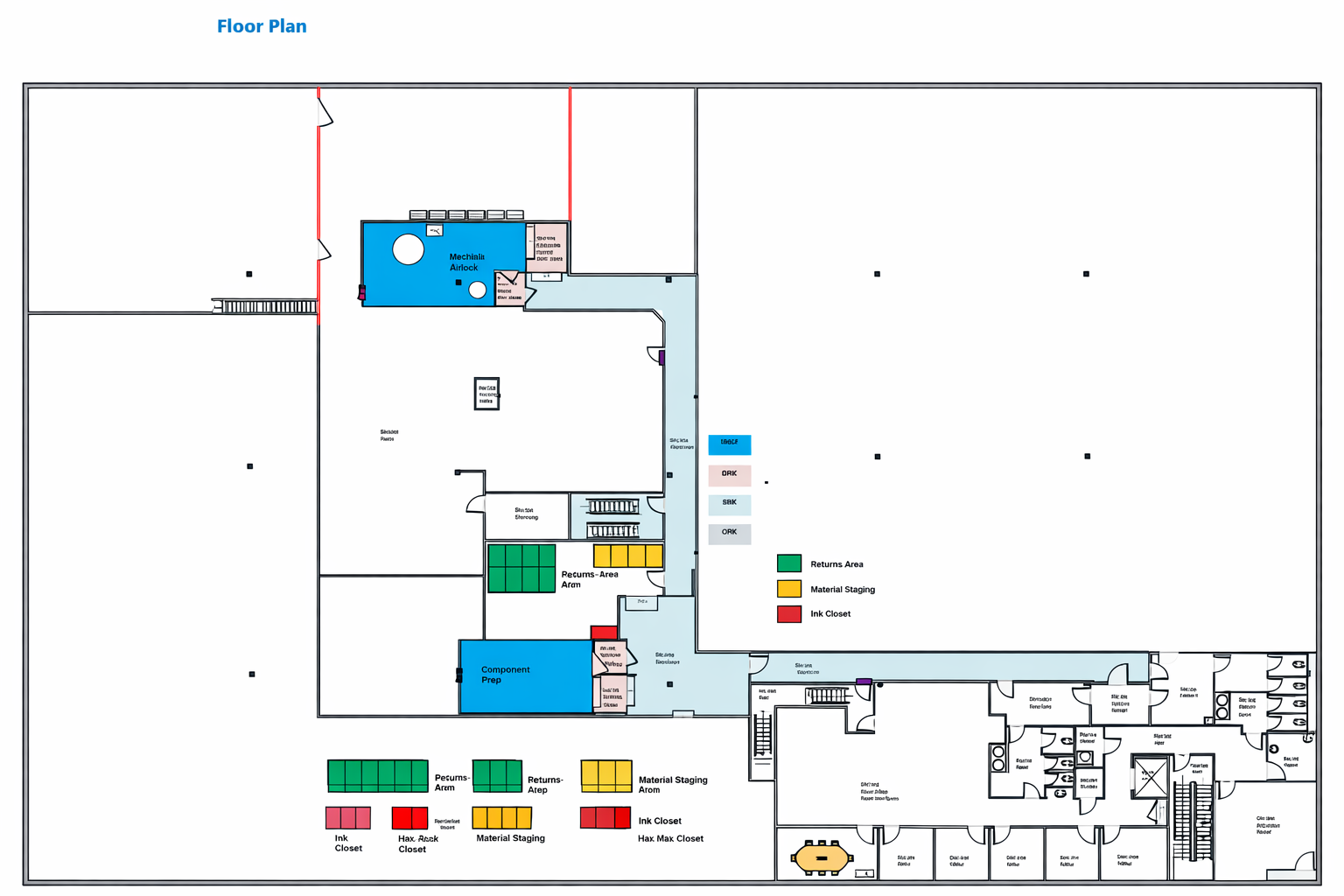
Rather than adapting legacy layouts to modern requirements, we started with the realities of sterile injectable manufacturing today and built forward. The result is a platform where quality, automation, and expansion are integrated rather than layered on.
Design choices were driven by
- Contamination control
- Operational flow
- Inspection readiness
- Long-term scalability
This approach reduces friction, complexity, and downstream risk.
A Platform, Not a Single Line
Bespoke operates as a manufacturing platform rather than a collection of isolated rooms or lines. Core infrastructure, utilities, and systems were designed to support growth within the same footprint. Expansion does not require rethinking the foundation. It builds upon it. This allows programs to scale without disruption, relocation, or forced technology transfer.
Automation With Purpose
Automation at Bespoke exists to protect product integrity. Robotic aseptic operations, closed systems, and automated inspection reduce variability, minimize human intervention, and improve repeatability. Automation is applied where it meaningfully reduces risk, not where it adds complexity. This focus supports consistent execution under real-world conditions, not just ideal scenarios.
Integrated From Compounding to Final Presentation
Manufacturing at Bespoke is designed as a continuous, controlled flow. Compounding, aseptic fill and finish, inspection, and packaging are integrated within the platform to reduce handoffs, limit exposure, and maintain traceability throughout the process. This integration strengthens quality control and simplifies documentation while improving execution efficiency.

Built for Inspection Readiness
Inspection readiness is not a future state. It is a design requirement.
- Facility layout, material flows, documentation practices, and operational controls were all selected with regulatory scrutiny in mind.
- The platform supports clear visibility into how work is performed, how decisions are made, and how control is maintained.
- The facility does not rely on workarounds to appear compliant. It was built to be compliant.
Expansion Without Compromise
Growth is anticipated, not reacted to.
Our facility blueprint supports additional manufacturing suites and future capabilities within the existing footprint. Core utilities and infrastructure are already provisioned to support expansion.
This forward-looking design allows Bespoke to grow deliberately while preserving control, quality, and operational consistency.
Built in Las Vegas, Nevada for a Reason
The choice of Las Vegas, Nevada was intentional. The region offers logistical reach, operational stability, and an environment conducive to advanced manufacturing without the congestion and fragility of traditional coastal hubs.
- Reliable distribution
- Reduced disruption risk
- Sustained investment in infrastructure and talent
It is a foundation built for longevity.
~Bespoke Pharmaceutical

The Platform Advantage
What partners experience at Bespoke is not just a facility.
It is a system designed to perform under pressure.
Predictable execution
Designed workflows reduce variability and keep delivery consistent.
Defensible quality
Controls and documentation support confident regulatory scrutiny.
Scalable growth
Expansion is supported without sacrificing discipline or control.
A Facility That Serves the Mission
At Bespoke, the facility exists to serve the mission, not the other way around.
Every wall, system, and workflow supports our commitment to quality, accountability, and long-term program success.
Ready to See the Platform in Action?
If you are evaluating a manufacturing partner based on infrastructure that can grow with your program, we are ready to talk.
©2026 | Bespoke Pharmaceutical | All Rights Reserved.




